Practical information
What is PIPAC®?
PIPAC®* is an innovative technique for treating peritoneal carcinosis. *Pressurised Intra Peritoneal Aerosol Chemotherapy (PIPAC®)
It involves delivering a chemotherapy agent in aerosol form, under pressure, directly into a patient’s peritoneal cavity during laparoscopy using an injector.
PIPAC®
What is the procedure?
PIPAC® is a minimally invasive laparoscopic procedure lasting 45 minutes during which chemotherapy is administered under pressure as an aerosol. Chemotherapy is administered at a concentration 10 times lower than that used in systemic treatment, directly on the cancerous lesions. The combination of aerosol form and pressure improves pharmacology and dynamics, resulting in homogeneous intraperitoneal diffusion of the cytotoxic drugs administered. Nearly 100% of drugs administered by aerosol reach the target. Side effects are minimal. La PIPAC® is well tolerated and is performed on an outpatient basis in some countries.
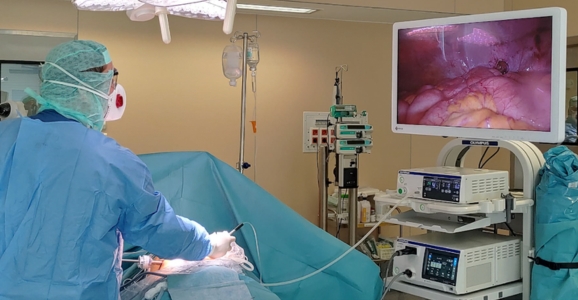
How effective is PIPAC®?
The current efficacy of PIPAC® is limited by the use of compounds that have not been developed for intraperitoneal administration. The choice of drug is largely arbitrary. Personalised therapy and sustained-release formulations offer the prospect of improved efficacy. [Source: website: https://capnopharm.com/]
How does a PIPAC® procedure work?
This technique is performed by conventional laparoscopy using 2 or 3 balloon trocars (to ensure a watertight seal). The first trocar, into which the optic is inserted, will be used for exsufflation at the end of the procedure. The second is designed to create abdominal overpressure by injecting CO2, and will house the nozzle for intraperitoneal injection of the chemotherapy. The third, which is optional, is used to assist the operator during the surgical procedures preceding the injection (viscerolysis, exploration, biopsies, etc.).
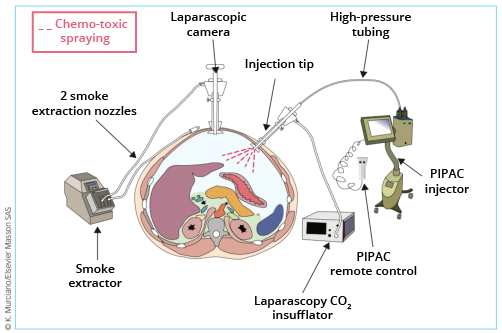
What are the indications?
La PIPAC® can be offered to patients with peritoneal carcinosis whether the carcinosis is of colorectal origin, gastric carcinosis, ovarian carcinosis or a rare disease such as peritoneal pseudomyxoma or peritoneal mesothelioma. This technique is offered to patients whose disease has been stabilised by the regular administration of systemic chemotherapy (intravenous) but who are unable to undergo Hyperthermic Intraperitoneal Chemotherapy (HIPEC) for various reasons.
It is repeated three times, six weeks apart, with the aim of limiting tumour progression, or even achieving a sufficient response to allow secondary complete cytoreduction surgery and HIPEC.
PIPAC®, a complementary treatment
PIPAC® does not compete with the standard treatment for peritoneal carcinosis, which is cytoreduction surgery combined with HIPEC. Rather, it appears to be a complementary treatment which, in the long term, could be offered to patients who are too fragile to withstand HIPEC, who have developed temporary resistance to conventional systemic chemotherapy, or who have a cancer that has spread too far to benefit from full surgery with HIPEC.
Several clinical studies have been carried out in Germany by the team led by Prof. Reymond, who designed this technique. One of them demonstrated a clinical and/or histological response in more than 60% of cases in patients with recurrent peritoneal carcinosis of gynaecological origin who were resistant to conventional chemotherapy.
A phase II study investigating the safety and efficacy of PIPAC® in patients with platinum-resistant relapsed ovarian cancer showed more than 70% clinical or histological responses, with no change in quality of life.
[Various sources: IGR + website: https://CHU Lyon/]
Model available:
CapnoPen® CP-001
Sterile, single-use endoscopic nebuliser designed by CapnoPharm for the medical administration of therapeutic substances in the form of pressurised aerosols into body cavities



