Original therapies & innovative technologies
Hyperthermic Intraperitoneal Chemotherapy: HIPEC
Hyperthermic Intraperitoneal Chemotherapy: a combination of radical cytoreduction, intraperitoneal chemotherapy and hyperthermia.
Hyperthermic intraperitoneal chemotherapy (HIPEC) was developed in the 1990s. The original indication was pseudomyxoma peritonei. Indications were subsequently extended to carcinosis of colorectal and appendiceal origin, as well as peritoneal mesothelioma, and more recently to carcinosis of ovarian origin.
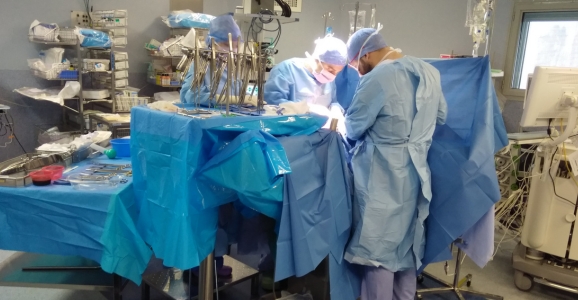
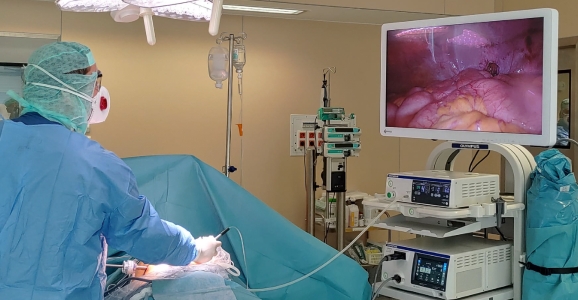
Pressurised Intraperitoneal Aerosolized Chemotherapy: PIPAC
In situations where carcinosis is very advanced from the outset, or if complete cytoreduction is not feasible PIPAC (Pressurised Intra Peritoneal Aerosol Chemotherapy) is a new therapeutic alternative which has been developing since 2010. This involves delivering chemotherapy intraperitoneally, in aerosolized and pressurized form to promote tissue penetration of the drugs. PIPAC sessions are performed every 6 to 8 weeks, combined with 2 courses of intravenous chemotherapy during this period.
Did you know?
All medical devices must be certified in accordance with strict regulations. In Europe, compliance with these regulations is evidenced by the issue of the medical CE mark which guarantees that the medical device meets the safety and clinical benefit requirements laid down.
Since May 2021, medical devices are subject to a new EU regulation 2017/745 (MDR: Medical Device Regulation).
By 26 May 2024, all medical devices must be marked CE MDR or have obtained an extension of their certificate following the implementation of an MDR approach by submitting an assessment request to a notified body.
In France, the quality and safety (materiovigilance, control, inspection) of all medical devices placed on the market are also monitored by the Agence nationale de sécurité du médicament et des produits de santé (ANSM).


