Lumbar SPROTTE® NEEDLE

Atraumatic needle for lumbar puncture and manometer to measure CSF pressure
The pioneer of Atraumatic Lumbar Puncture
- Description
- information
- benefits
- studies
- legal information
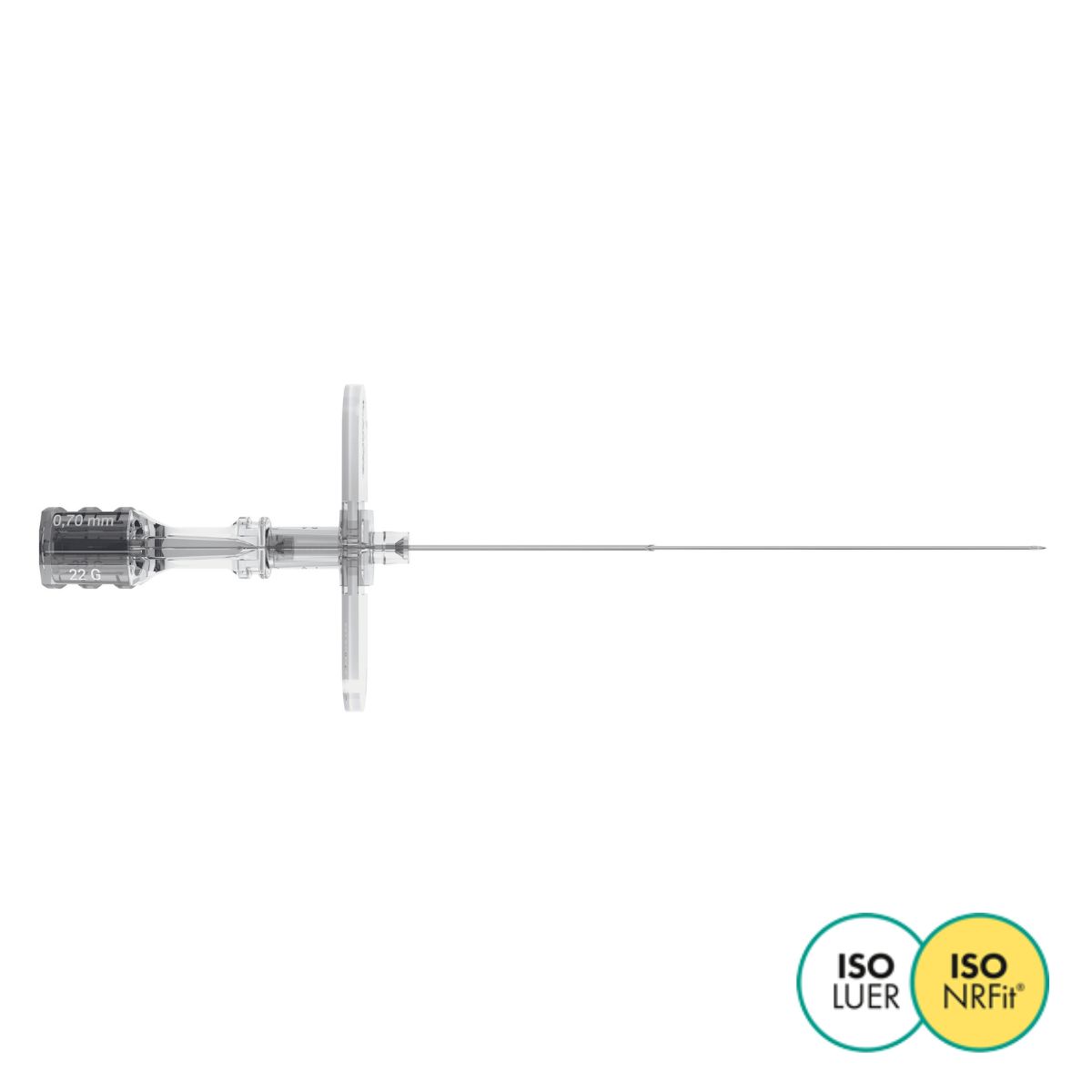
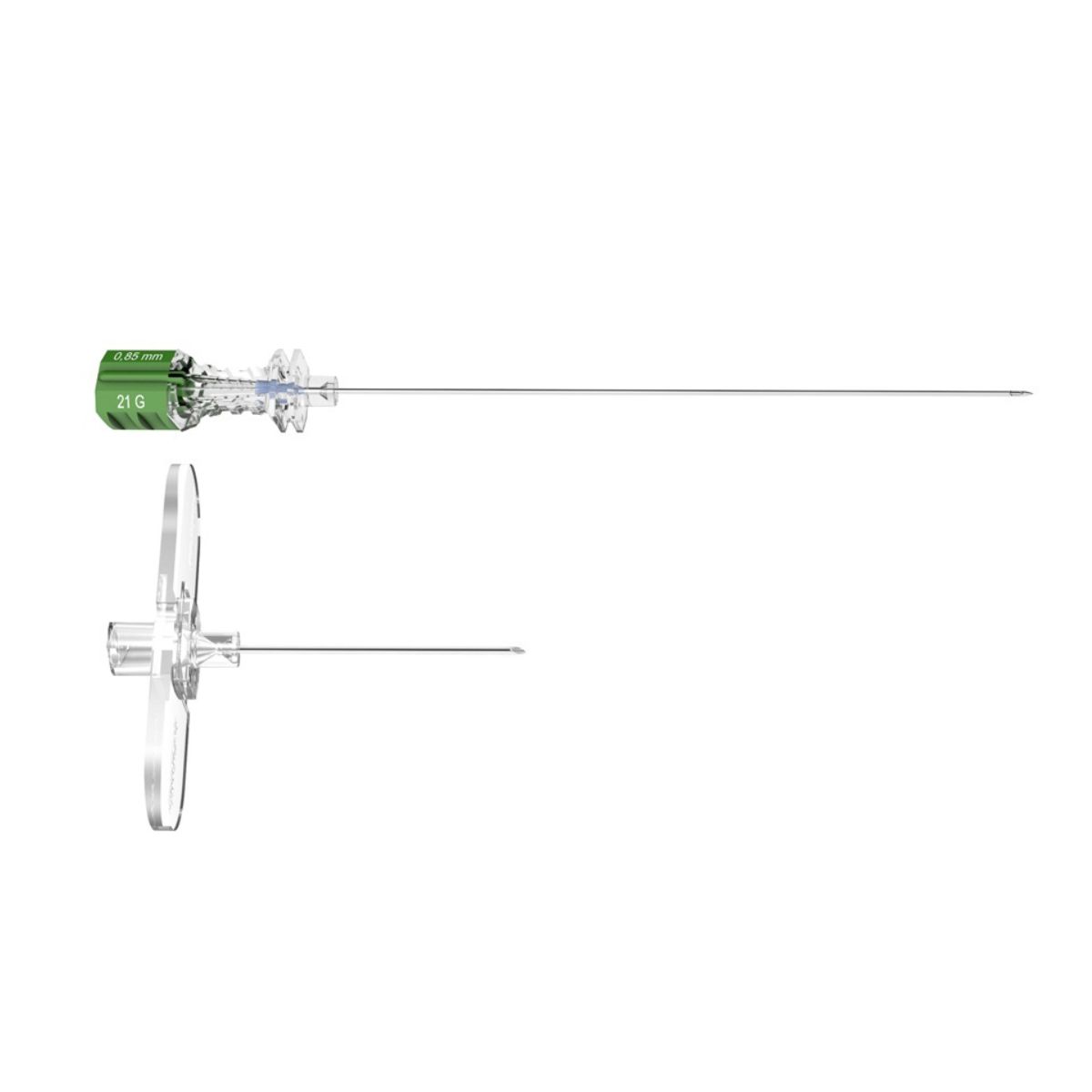
Invented in 1979 as a result of collaboration between Pajunk® and Professor Günter Sprotte, the Sprotte® needle is the first atraumatic needle for Lumbar Puncture. The secret of its success lies in the unique geometry of its tip and the architecture of its hub. This design, developed specifically for lumbar puncture, enables atraumatic puncture of the various membranes and optimises CSF flow, while reducing the incidence of post dural puncture headaches (PDPH).
The Sprotte® needle guarantees patients and users functionality, safety and reliability, as well as a reduction in the incidence of post dural puncture headaches. This translates into an economic gain linked to the absence of costs associated with the management of post-lumbar puncture syndrome (re-hospitalisation, bloody patch, etc.).
With the Sprotte® lumbar needle and Pressure manometer, Pajunk® provides a complete set of equipment for diagnostic CSF sampling and pressure measurement.
- The Sprotte® needle for Lumbar Puncture is available in lengths from 90 to 120 mm and diameters from 20 to 22 G.
- Available with LUER or NRFit connectors.
- The Pajunk® spinal manometer allows measurements up to 54 cm H20 (with additional extension tube) and is available in NRFit®.
- Improves safety of procedures
- Earlier detection of cerebrospinal fluid (CSF) thanks to its magnified hub
- Enables rapid detection of the smallest quantities of CSF
- Minimises the risk of PDPH (Post Dural Puncture Headache) thanks to the atraumatic tip
- Minimises the risk of tissue coring thanks to the needle's lateral eye and a perfectly adapted metal stylet
- Increases efficacity, reduces the cost of treating PDPH
- Facet tip introducer with diameter and length adapted to each needle size, reducing the risk of damaging the Sprotte® needle during insertion
- Davis,A.;Dobson,R.;Kaninia,S.;Giovannoni,G.;Schmierer,K.(2016): Atraumatic needles for lumbar puncture: why haven't neurologists changed? In Practical neurology 16(1),pp. 1822. DOI: 10.1136/practneurol-2014-001055.
- Engedal,Thorbjørn S.;Ørding,Helle;Vilholm,Ole Jakob(2015): Changing the needle for lumbar punctures: results from a prospective study. In Clinical neurology and neurosurgery 130,pp. 7479. DOI: 10.1016/j.clineuro.2014.12.020.
- Tung,C. E.;Yuen,T. S.;Lansberg,M. G.(2012): Cost comparison between the atraumatic and cutting lumbar puncture needles. In Neurology 78,pp. 109113.
- Arevalo-Rodriguez,Ingrid;Muñoz,Luis;Godoy-Casasbuenas,Natalia;Ciapponi,Agustín;Arevalo,Jimmy J.;Boogaard,Sabine;Roqué I Figuls,Marta(2017): Needle gauge and tip designs for preventing post-dural puncture headache(PDPH). In The Cochrane database of systematic reviews 4,CD010807. DOI: 10.1002/14651858.CD010807.pub2.
- McLaughlin,Colleen A.;Hockenberry,Marilyn J.;Kurtzberg,Joanne;Hueckel,Rémi;Martin,Paul L.;Docherty,Sharron L.(2014): Standardization of health care provider competencies for intrathecal access procedures. In Journal of pediatric oncology nursing: official journal of the Association of Pediatric Oncology Nurses 31(6),pp. 304316. DOI: 10.1177/1043454214543019.
- Nath,Siddharth;Koziarz,Alex;Badhiwala,Jetan H.;Alhazzani,Waleed;Jaeschke,Roman;Sharma,Sunjay et al.(2018): Atraumatic versus conventional lumbar puncture needles: a systematic review and meta-analysis. In The Lancet 391(10126),pp. 11971204. DOI: 10.1016/S0140-6736(17)32451-0.
- Rochwerg,Bram;Almenawer,Saleh A.;Siemieniuk,Reed A. C.;Vandvik,Per Olav;Agoritsas,Thomas;Lytvyn,Lyubov et al.(2018): Atraumatic(pencil-point)versus conventional needles for lumbar puncture: a clinical practice guideline. In BMJ,k1920. DOI: 10.1136/bmj.k1920.
- Zhang,Yi C.;Chandler,Alexander J.;Kagetsu,Nolan J.(2014): Technical compliance to standard guidelines for lumbar puncture and myelography: survey of academic neuroradiology attendings and fellows. In Academic radiology 21(5),pp. 612616. DOI: 10.1016/j.acra.2014.01.021.
- Bellamkonda,Venkatesh R.;Wright,Thomas C.;Lohse,Christine M.;Keaveny,Virginia R.;Funk,Eric C.;Olson,Michael D.;Laack,Torrey A.(2017): Effect of spinal needle characteristics on measurement of spinal canal opening pressure. In The American journal of emergency medicine 35(5),pp. 769772. DOI: 10.1016/j.ajem.2017.01.047.
- Puolakka,R.;Andersson,L. C.;Rosenberg,H.(2000): Microscopic Analysis of Three Different Spinal Needle Tips After Experimental Subarachnoid Puncture. In Regional Anesthesia and Pain Medicine 25(2),pp. 163169.
- Lavi R.,Rowe J.M.,Avivi I. Lumbar Puncture. It Is Time to Change the Needle,Eur Neurol,2010;64:108113
Description
Invented in 1979 as a result of collaboration between Pajunk® and Professor Günter Sprotte, the Sprotte® needle is the first atraumatic needle for Lumbar Puncture. The secret of its success lies in the unique geometry of its tip and the architecture of its hub. This design, developed specifically for lumbar puncture, enables atraumatic puncture of the various membranes and optimises CSF flow, while reducing the incidence of post dural puncture headaches (PDPH).
The Sprotte® needle guarantees patients and users functionality, safety and reliability, as well as a reduction in the incidence of post dural puncture headaches. This translates into an economic gain linked to the absence of costs associated with the management of post-lumbar puncture syndrome (re-hospitalisation, bloody patch, etc.).
With the Sprotte® lumbar needle and Pressure manometer, Pajunk® provides a complete set of equipment for diagnostic CSF sampling and pressure measurement.
Additional information
| Information | <ul><li>The Sprotte® needle for Lumbar Puncture is available in lengths from 90 to 120 mm and diameters from 20 to 22 G.</li><li>Available with LUER or NRFit connectors.</li><li>The Pajunk® spinal manometer allows measurements up to 54 cm H20 (with additional extension tube) and is available in NRFit®.</li></ul> |
|---|---|
| Benefits | <ul><li> Improves safety of procedures</li><li>Earlier detection of cerebrospinal fluid (CSF) thanks to its magnified hub</li><li>Enables rapid detection of the smallest quantities of CSF</li><li>Minimises the risk of PDPH (Post Dural Puncture Headache) thanks to the atraumatic tip</li><li>Minimises the risk of tissue coring thanks to the needle's lateral eye and a perfectly adapted metal stylet </li><li>Increases efficacity, reduces the cost of treating PDPH</li><li>Facet tip introducer with diameter and length adapted to each needle size, reducing the risk of damaging the Sprotte® needle during insertion</li></ul> |
| Studies | <ol start="1"><li>Davis,A.;Dobson,R.;Kaninia,S.;Giovannoni,G.;Schmierer,K.(2016): Atraumatic needles for lumbar puncture: why haven't neurologists changed? In Practical neurology 16(1),pp. 1822. DOI: 10.1136/practneurol-2014-001055.</li><li>Engedal,Thorbjørn S.;Ørding,Helle;Vilholm,Ole Jakob(2015): Changing the needle for lumbar punctures: results from a prospective study. In Clinical neurology and neurosurgery 130,pp. 7479. DOI: 10.1016/j.clineuro.2014.12.020.</li><li>Tung,C. E.;Yuen,T. S.;Lansberg,M. G.(2012): Cost comparison between the atraumatic and cutting lumbar puncture needles. In Neurology 78,pp. 109113.</li><li>Arevalo-Rodriguez,Ingrid;Muñoz,Luis;Godoy-Casasbuenas,Natalia;Ciapponi,Agustín;Arevalo,Jimmy J.;Boogaard,Sabine;Roqué I Figuls,Marta(2017): Needle gauge and tip designs for preventing post-dural puncture headache(PDPH). In The Cochrane database of systematic reviews 4,CD010807. DOI: 10.1002/14651858.CD010807.pub2.</li><li>McLaughlin,Colleen A.;Hockenberry,Marilyn J.;Kurtzberg,Joanne;Hueckel,Rémi;Martin,Paul L.;Docherty,Sharron L.(2014): Standardization of health care provider competencies for intrathecal access procedures. In Journal of pediatric oncology nursing: official journal of the Association of Pediatric Oncology Nurses 31(6),pp. 304316. DOI: 10.1177/1043454214543019.</li><li>Nath,Siddharth;Koziarz,Alex;Badhiwala,Jetan H.;Alhazzani,Waleed;Jaeschke,Roman;Sharma,Sunjay et al.(2018): Atraumatic versus conventional lumbar puncture needles: a systematic review and meta-analysis. In The Lancet 391(10126),pp. 11971204. DOI: 10.1016/S0140-6736(17)32451-0.</li><li>Rochwerg,Bram;Almenawer,Saleh A.;Siemieniuk,Reed A. C.;Vandvik,Per Olav;Agoritsas,Thomas;Lytvyn,Lyubov et al.(2018): Atraumatic(pencil-point)versus conventional needles for lumbar puncture: a clinical practice guideline. In BMJ,k1920. DOI: 10.1136/bmj.k1920.</li><li>Zhang,Yi C.;Chandler,Alexander J.;Kagetsu,Nolan J.(2014): Technical compliance to standard guidelines for lumbar puncture and myelography: survey of academic neuroradiology attendings and fellows. In Academic radiology 21(5),pp. 612616. DOI: 10.1016/j.acra.2014.01.021.</li><li>Bellamkonda,Venkatesh R.;Wright,Thomas C.;Lohse,Christine M.;Keaveny,Virginia R.;Funk,Eric C.;Olson,Michael D.;Laack,Torrey A.(2017): Effect of spinal needle characteristics on measurement of spinal canal opening pressure. In The American journal of emergency medicine 35(5),pp. 769772. DOI: 10.1016/j.ajem.2017.01.047.</li><li>Puolakka,R.;Andersson,L. C.;Rosenberg,H.(2000): Microscopic Analysis of Three Different Spinal Needle Tips After Experimental Subarachnoid Puncture. In Regional Anesthesia and Pain Medicine 25(2),pp. 163169.</li><li>Lavi R.,Rowe J.M.,Avivi I. Lumbar Puncture. It Is Time to Change the Needle,Eur Neurol,2010;64:108113</li></ol> |
| Legal information | Sprotte: Atraumatic needle with Sprotte tip for lumbar puncture. Class III medical device. CE0124. Certification body: DEKRA. Manufactured by Pajunk GmbH. Not included in the list of reimbursable products and services provided for in article L165-1 of the French Social Security Code. Read the instructions carefully before use. Manometer: Spinal manometer with 3-way valve with LUER or NRFit connection. Class Is medical device. CE0124. Certification body: DEKRA. Manufactured by Pajunk GmbH. Not included in the list of reimbursable products and services provided for in article L165-1 of the French Social Security Code. Read the instructions carefully before use. |
| Bandeau | <img src="https://gamida.fr/wp-content/uploads/2025/03/set-personnalise-2-EN.png"> |
| Traduction_FR | 5178 |
| Fabricant | <img src="https://gamida.fr/wp-content/uploads/2024/02/logo_pajunk.jpg"> |
DID YOU KNOW?