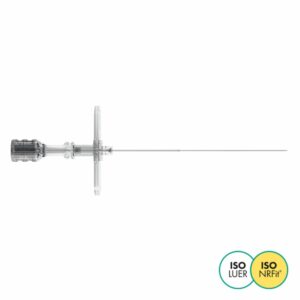
Lumbar puncture
Essential medical procedure
Lumbar puncture (LP) is an essential and very common medical procedure. It can give rise to adverse events, which are rarely serious, but the incidence of which can be reduced by applying simple measures.
Post-lumbar puncture syndrome (PLPS) is the most frequent adverse event. Secondary to a persistent leak of cerebrospinal fluid (CSF), it manifests itself as an orthostatic headache, usually appearing 2 to 4 days after the procedure.
Sprotte® needles
The first atraumatic needles for lumbar puncture
“Pajunk®, 40 years of experience in lumbar puncture.
Invented in 1979, the result of a collaboration between Pajunk® and Professor Sprotte, the Sprotte® needle is the first atraumatic needle for lumbar puncture.
The secret of its success lies in the unique geometry of its tip and the architecture of its base.
This design, developed specifically for lumbar puncture, enables atraumatic puncture of the various membranes and optimises cerebrospinal fluid (CSF) reflux while reducing the incidence of post dural puncture headaches (PDPH).

Prof. Günter Sprotte
Find out more
Sprotte® Needle and Manometer
The Sprotte® needle guarantees patients and users functionality, safety and reliability, as well as a reduction in the incidence of post dural puncture headaches.
Adverse advents of lumbar puncture
HAS recommendations
The HAS published recommendations in June 2019 for “the prevention and management of adverse events that may occur after lumbar puncture”.
It is recommended that a blunt atraumatic needle and introducer be used, whatever the indication for lumbar puncture, in both adults and paediatrics.
It is recommended that doctors be trained in LP procedure, as well as in the use of atraumatic needles with introducers, and that they be supported as often as necessary when performing the first LP on the patient.
Adherence to procedural rules and the use of atraumatic needles significantly reduce the incidence of adverse events and the use of blood patches
To find out more
How to perform the procedure
Setting up the patient
- The choice of sitting or lying position is at the doctor and patient’s discretion
Determining the entry site
- The correct entry sites are the L3-L4, L4-L5 and L5-S1 interspinous spaces.
Asepsis
- The rules of surgical asepsis must be strictly observed:
- for the patient: two-stage skin disinfection with an antiseptic (alcoholic antiseptic) and use of a sterile drape;
- for the doctor: hand disinfection (hydro-alcoholic solution), face mask and sterile gloves
Local anaesthesia
- Patients may be offered a local anaesthetic patch outside the emergency room (within 1 hour). In foreseeable cases where entry is difficult, a local anaesthetic may be considered.
Performing needle entry
- Regardless of the indication for LP, the technique and procedures remain the same. It is recommended that entry is with a blunt atraumatic needle with a maximum diameter of 22 Gauge (black colour code). It is essential to use the introducer supplied with the needle to penetrate the skin. Doctors should be trained in the use of atraumatic needles with introducers.
Reinserting the mandrel
- It is advisable to completely reinsert the mandrel into the needle before removing it.
Sampling
- The incidence of immediate post-LP syndromes increases for volumes greater than 30 ml.
Preventing accidents involving exposure to blood
- LP is an invasive procedure with a risk of blood exposure, and needles must be collected in a special container provided for this purpose.
Did you know?
Atraumatic needles for lumbar puncture are covered by the new NRFit® standard.
Pajunk’s atraumatic Sprotte® needles are all available with this new NRFit® connector.